Ionic Material
Ionic material is classified as any material that both dissolves in water and produces positive ions (cations) and negative ions (anions). There are two important processes to consider: solubility and ionization. Before a substance can produce ions, it must dissolve in water. All compounds have a characteristic “solubility product,” which is associated with the extent of saturation of the material in a water solution. A highly soluble material, for example, is sodium chloride (NaCl), common table salt. It readily dissolves in water, producing a sodium ion with a +1 charge and a chloride ion with a -1 charge:
NaCl + Water → Na+ + Cl–
It is important to consider that even very soluble sodium chloride can reach a saturation limit; it becomes particulate matter when its saturation value is exceeded. To verify, one can slowly pour salt into a glass of water. During this procedure, it will become obvious that the saturation point is reached when the sodium chloride no longer dissolves in the water, but settles to the bottom of the glass. Compounds such as calcium carbonate (CaCO3) and magnesium carbonate (MgCO3) are much less soluble in water than sodium chloride. In general, salts of monovalent, low molecular weight cations (e.g., Na) and monovalent, low molecular weight anions (e.g., Cl) are relatively soluble.
It would be inappropriate to conclude a discussion of solubility and subsequent ionization in water without addressing the relationship of ionic removal to the “dissolving” process. This concept can be explained by relating it to a water purification unit operation water softening with a regenerant salt storage tank. When wet salt storage is employed within the brine tank, salt pellets remain below the level of water. When the water softener is regenerated, saturated salt solution is withdrawn from the salt storage tank. The saturated brine solution is diluted with water and regenerates the cation resin in the water softener. Prior to the next regeneration cycle, makeup water is introduced into the salt storage tank. Previously non-dissolved salt dissolves in the makeup water. The salt level in the salt storage tank decreases each time the unit is regenerated. Thus, increased solubility occurs by dilution of the water solution in the salt tank. Ions are produced when certain compounds are added to water and dissolved to produce an electrically neutral solution of positive and negative ions
The ionic material produced can be related to a single atom (e.g. Na+) or a radical (e.g. SO4–). For purposes of defining ions for water purification, it is relatively unimportant to refer to molecular weights and the associated charge (monovalent, divalent, etc.). Obviously, the ability of a compound to produce ions is extremely important to certain unit operations (e.g. ion exchange). As ions enter a water solution, the ability to conduct an electrical current increases. An indication of the concentration of ions in a water solution is determined by measuring the conductance or resistance to an applied electric current associated with a probe immersed in a water solution containing the ions. Prior to discussing the measurement of the ionic strength of a water solution, it is important to indicate that the equivalent conductance of all positive and negative ions is, in general, approximately equal, with the exception of the hydronium (H3O+) and hydroxyl (OH–) ions. This important ionic property, for water solutions with relatively neutral pH values, can be used to approximate the total dissolved solid (TDS) level of a solution.
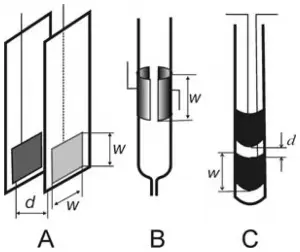
While most conductivity or resistivity cells used with water purification systems have a complex configuration, the basic principles associated with the measurement can be explained by referring to a classical dip-type cell, as opposed to a flow-through cell. Within the cell, two parallel plates are positioned. The effective area of each plate, W, is expressed in cm2. The length (d) between the plates is expressed in cm. A cell constant is generated by dividing the length between the plates by the effective area of each plate (d/W). The conductance or resistance between the two plates is measured. The specific conductance, or conductivity, is determined by multiplying the measured conductance by the cell constant. Conversely, the specific resistance, or resistivity, is determined by dividing the resistance by the cell constant. As a result, conductivity or resistivity cannot be expressed without a dimensional term. For the vast majority of water purification applications, conductivity is expressed a micro-siemens (µS)/cm. Resistivity is expressed as Ω-cm, kΩ-cm, or, for high purity water, MΩ-cm (megaohms-cm). Since water temperature increases the mobility of ions, conductivity and resistivity should reference a measurement temperature such as 250C or be corrected to 250C by using appropriate correction curves. The units for conductivity and resistivity are direct reciprocals of each other. The maximum obtainable resistivity for theoretically ion-free water is 18.2 MΩ-cm (0.055 µS/cm conductivity) at 250C.
From a theoretical standpoint, the conductivity of a solution can be calculated by using the equivalent conductance of individual ions and multiplying by the concentration of the specific ions. An approximation of the TDS level of ions in solution can be obtained by multiplying the conductivity (at 250C) by 0.5. Again, this calculation only provides an estimate of the TDS value.
The level of ionic material in water determines the purity of the water. For sodium chloride in water, the solubility and direct ionization characteristics were presented. For pharmaceutical water applications, one can assume that the sodium chloride that dissolves in solution is ionized as sodium and chloride ions. Some salts, acids, and bases do not completely ionize. These materials will dissolve (based on their solubility product), producing, in a simplified nomenclature, a unionized portion of the original material and an ionized portion of the material. Examples of weakly ionized salts, existing in equilibrium with a dissolved unionized portion.
The degree of ionization for each equilibrium equation is determined by an ionization constant. The larger the ionization constant, the greater the percentage of ions present when compared with the unionized portion. The lower the ionization constant, the greater the percentage of unionized material compared to ionized material. The kinetics of the equilibrium reaction, particularly as it relates to gaseous components, is a very critical factor in determining the product water purity from a single or double pass reverse osmosis (RO) unit. Subsequently, it is very important to remember that certain materials are weakly ionized when evaluating the use of RO for particular applications.
Some salts, which are weakly ionized, are referred to as acid salts or basic salts. Adding sodium chloride to water has no effect on the pH of the solution. However, if Trisodium Phosphate (Na3PO4) is added to water, a noticeable increase in pH will occur.
Basic salts consist of a strongly ionized anion and weakly ionized cation. An acid salt consists of a strongly ionized cation and weakly ionized anion. In general, strongly and weekly ionized ions relate to their associated acid or base.
For water purification purposes, a general understanding of these phenomena is important. Since raw water supplies consist of both weakly and strongly ionized cations and anions, the resulting pH is affected by numerous equilibrium. It is not so much important to know the nature of the equilibrium involved or the equilibrium (ionization) constants. However, it is important to recognize that the effect of this situation is exhibited by a pH value that can be either slightly acidic or slightly basic.